Childhood Central Nervous System Atypical Teratoid/Rhabdoid Tumor Treatment (PDQ®): Treatment - Health Professional Information
Childhood Central Nervous System Atypical Teratoid/Rhabdoid Tumor Treatment (PDQ®): Treatment - Health Professional Information [NCI]Skip to the navigationGeneral Information About Childhood Central Nervous System (CNS) Atypical Teratoid / Rhabdoid TumorDramatic improvements in survival have been achieved for children and adolescents with cancer. Between 1975 and 2010, childhood cancer mortality decreased by more than 50%.[1] Childhood and adolescent cancer survivors require close follow-up because cancer therapy side effects may persist or develop months or years after treatment. (Refer to the PDQ summary on Late Effects of Treatment for Childhood Cancer for specific information about the incidence, type, and monitoring of late effects in childhood and adolescent cancer survivors.) Primary brain tumors are a diverse group of diseases that together constitute the most common solid tumor of childhood. The PDQ childhood brain tumor treatment summaries are primarily organized according to the World Health Organization classification of nervous system tumors.[2,3] Brain tumors are classified according to histology, but immunohistochemical analysis, cytogenetic and molecular genetic findings, and measures of mitotic activity are increasingly used in tumor diagnosis and classification. Tumor location and extent of spread are important factors that affect treatment and prognosis. For a full description of the classification of nervous system tumors and a link to the corresponding treatment summary for each type of brain tumor, refer to the PDQ summary on Childhood Brain and Spinal Cord Tumors Treatment Overview. Central nervous system (CNS) atypical teratoid/rhabdoid tumor (AT/RT) is a rare, clinically aggressive tumor that most often affects children aged 3 years and younger but can occur in older children and adults. About one-half of AT/RTs arise in the posterior fossa. The diagnostic evaluation includes magnetic resonance imaging (MRI) of the neuraxis and lumbar cerebrospinal fluid examination. AT/RT has been linked to somatic and germline mutations of SMARCB1, a tumor suppressor gene. There is no current standard treatment for children with AT/RT. Multimodal treatment consisting of surgery, chemotherapy, and radiation therapy is under evaluation. Based on present biologic understanding, AT/RT is part of a larger family of rhabdoid tumors. In this summary, the term AT/RT refers to CNS tumors only and the term rhabdoid tumor reflects the possibility of both CNS and non-CNS tumors. Unless specifically noted in the text, this summary is referring to CNS AT/RT. Incidence The exact incidence of childhood CNS AT/RT is difficult to determine because the tumor has been widely recognized for only the last decade. - In two North American prospective studies performed by the Children's Cancer Group and the Pediatric Oncology Group for children aged 3 years or younger at diagnosis, retrospective review disclosed that approximately 10% of children with brain tumors had AT/RTs.[4]
- A Taiwanese study found that AT/RTs account for 26% of primitive or embryonal tumors in children younger than 3 years.[5]
- The Austrian Brain Tumor Registry, conducted between 1996 and 2006, confirmed that AT/RTs represent the sixth most common malignant brain tumor among 311 newly diagnosed children (6.1%), with a peak incidence during the first 2 years of life.[6]
The incidence in older patients is unknown. However, in the Central Nervous System Atypical Teratoid/Rhabdoid Tumor Registry (AT/RT Registry), 12 (29%) of the 42 patients were older than 36 months at the time of diagnosis.[7] Anatomy
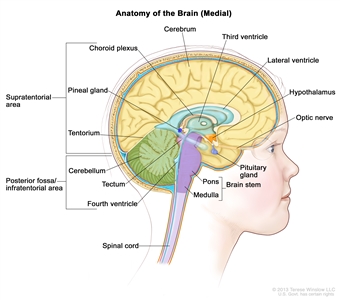
Anatomy of the inside of the brain, showing the pineal and pituitary glands, optic nerve, ventricles (with cerebrospinal fluid shown in blue), and other parts of the brain. The tentorium separates the cerebrum from the cerebellum. The infratentorium (posterior fossa) is the region below the tentorium that contains the brain stem, cerebellum, and fourth ventricle. The supratentorium is the region above the tentorium and denotes the region that contains the cerebrum.
Clinical Presentation Childhood AT/RT is a clinically aggressive tumor that primarily occurs in children younger than 3 years, but it also can occur in older children and has been reported in adults.[8,9] In about one-half of patients, the tumor is located in the posterior fossa, although it can occur anywhere in the CNS.[4] Tumors of the posterior fossa may occur in the cerebellopontine angle or more midline. Involvement of individual cranial nerves has also been noted. Because AT/RT grows rapidly, patients typically have a fairly short history of progressive symptoms, measured in days to weeks. Signs and symptoms are dependent on tumor location. Young patients with posterior fossa tumors usually present with symptoms related to hydrocephalus, including the following: - Early morning headaches.
- Vomiting.
- Lethargy.
They may also develop ataxia or regression of motor skills. Data from the AT/RT Registry suggest that approximately 20% of patients present with disseminated disease.[7] Dissemination is typically through leptomeningeal pathways seeding the spine and other areas of the brain. There are also reports of rare patients with synchronous renal rhabdoid tumor and CNS AT/RT.[10] Diagnostic Evaluation All patients with suspected childhood AT/RT should have MRI of the brain and spine. Unless medically contraindicated, patients should also have lumbar cerebrospinal fluid inspected for evidence of tumor. Patients may also undergo renal ultrasound to detect synchronous tumors. AT/RT cannot be reliably distinguished from other malignant brain tumors based on clinical history or radiographic evaluation. Surgery is necessary to obtain tissue and confirm the diagnosis of AT/RT. Immunostaining for loss of SMARCB1 (INI1, hSNF5) protein expression is used to confirm the diagnosis, especially in children younger than 3 years.[11] Prognosis Prognostic factors that affect survival for AT/RTs are not fully delineated. Known factors associated with a poor outcome include the following: - Germline mutation.[12]
- Age younger than 2 years.[13]
- Metastases at diagnosis.[13]
- Subtotal resection.[14]
Most published information on outcomes for patients with AT/RT is based on small series and is retrospective in nature. Initial retrospective studies reported an average survival from diagnosis of only about 12 months.[4,8,14,15,16] In a retrospective report, 2-year overall survival was better for patients who underwent a gross-total resection than for those who had a subtotal resection. However, in this study the effect of radiation therapy on survival was less clear.[14] There are reports of long-term survivors.[17] Notably, improved survival has been reported for those receiving intensive multimodal therapy. - Children aged 3 years and older who received postoperative craniospinal irradiation and high-dose, alkylator-based chemotherapy had improved survival compared with those younger than 3 years with AT/RT. In this report, the incidence of leptomeningeal metastases was also higher in the infant group of patients.[18]
- In one prospective study of 25 children with AT/RT who received intensive multimodal therapy, including radiation and intrathecal chemotherapy, the reported 2-year progression-free survival rate was 53%, and the overall survival rate was 70%.[19]
References:
-
Smith MA, Altekruse SF, Adamson PC, et al.: Declining childhood and adolescent cancer mortality. Cancer 120 (16): 2497-506, 2014.
-
Louis DN, Ohgaki H, Wiestler OD, et al., eds.: WHO Classification of Tumours of the Central Nervous System. 4th ed. Lyon, France: IARC Press, 2007.
-
Louis DN, Ohgaki H, Wiestler OD, et al.: The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114 (2): 97-109, 2007.
-
Packer RJ, Biegel JA, Blaney S, et al.: Atypical teratoid/rhabdoid tumor of the central nervous system: report on workshop. J Pediatr Hematol Oncol 24 (5): 337-42, 2002 Jun-Jul.
-
Ho DM, Hsu CY, Wong TT, et al.: Atypical teratoid/rhabdoid tumor of the central nervous system: a comparative study with primitive neuroectodermal tumor/medulloblastoma. Acta Neuropathol 99 (5): 482-8, 2000.
-
Woehrer A, Slavc I, Waldhoer T, et al.: Incidence of atypical teratoid/rhabdoid tumors in children: a population-based study by the Austrian Brain Tumor Registry, 1996-2006. Cancer 116 (24): 5725-32, 2010.
-
Hilden JM, Meerbaum S, Burger P, et al.: Central nervous system atypical teratoid/rhabdoid tumor: results of therapy in children enrolled in a registry. J Clin Oncol 22 (14): 2877-84, 2004.
-
Burger PC, Yu IT, Tihan T, et al.: Atypical teratoid/rhabdoid tumor of the central nervous system: a highly malignant tumor of infancy and childhood frequently mistaken for medulloblastoma: a Pediatric Oncology Group study. Am J Surg Pathol 22 (9): 1083-92, 1998.
-
Lutterbach J, Liegibel J, Koch D, et al.: Atypical teratoid/rhabdoid tumors in adult patients: case report and review of the literature. J Neurooncol 52 (1): 49-56, 2001.
-
Biegel JA, Fogelgren B, Wainwright LM, et al.: Germline INI1 mutation in a patient with a central nervous system atypical teratoid tumor and renal rhabdoid tumor. Genes Chromosomes Cancer 28 (1): 31-7, 2000.
-
Bruggers CS, Bleyl SB, Pysher T, et al.: Clinicopathologic comparison of familial versus sporadic atypical teratoid/rhabdoid tumors (AT/RT) of the central nervous system. Pediatr Blood Cancer 56 (7): 1026-31, 2011.
-
Kordes U, Gesk S, Frühwald MC, et al.: Clinical and molecular features in patients with atypical teratoid rhabdoid tumor or malignant rhabdoid tumor. Genes Chromosomes Cancer 49 (2): 176-81, 2010.
-
Dufour C, Beaugrand A, Le Deley MC, et al.: Clinicopathologic prognostic factors in childhood atypical teratoid and rhabdoid tumor of the central nervous system: a multicenter study. Cancer 118 (15): 3812-21, 2012.
-
Lafay-Cousin L, Hawkins C, Carret AS, et al.: Central nervous system atypical teratoid rhabdoid tumours: the Canadian Paediatric Brain Tumour Consortium experience. Eur J Cancer 48 (3): 353-9, 2012.
-
Rorke LB, Packer RJ, Biegel JA: Central nervous system atypical teratoid/rhabdoid tumors of infancy and childhood: definition of an entity. J Neurosurg 85 (1): 56-65, 1996.
-
Athale UH, Duckworth J, Odame I, et al.: Childhood atypical teratoid rhabdoid tumor of the central nervous system: a meta-analysis of observational studies. J Pediatr Hematol Oncol 31 (9): 651-63, 2009.
-
Olson TA, Bayar E, Kosnik E, et al.: Successful treatment of disseminated central nervous system malignant rhabdoid tumor. J Pediatr Hematol Oncol 17 (1): 71-5, 1995.
-
Tekautz TM, Fuller CE, Blaney S, et al.: Atypical teratoid/rhabdoid tumors (ATRT): improved survival in children 3 years of age and older with radiation therapy and high-dose alkylator-based chemotherapy. J Clin Oncol 23 (7): 1491-9, 2005.
-
Chi SN, Zimmerman MA, Yao X, et al.: Intensive multimodality treatment for children with newly diagnosed CNS atypical teratoid rhabdoid tumor. J Clin Oncol 27 (3): 385-9, 2009.
Tumor Biology of Childhood CNS Atypical Teratoid / Rhabdoid TumorChildhood central nervous system (CNS) atypical teratoid/rhabdoid tumor (AT/RT) was first described as a discrete clinical entity in 1987 [1] based on its distinctive pathologic and genetic characteristics. Before then, it was most often classified as a medulloblastoma, primitive neuroectodermal tumor, or choroid plexus carcinoma. The World Health Organization (WHO) began classifying AT/RT as an embryonal grade IV neoplasm in 1993.[2] Histologically, classic AT/RT is morphologically heterogeneous, typically containing sheets of large epithelioid cells with abundant eosinophilic cytoplasm and scattered rhabdoid cells, most often with accompanying components of primitive neuroectodermal cells (small round blue cells), mesenchymal cells, and/or glial cells.[3] Immunohistochemical staining for epithelial markers (cytokeratin or epithelial membrane antigen), glial fibrillary acidic protein, synaptophysin (or neurofilament), and smooth muscle (desmin) may help to identify the heterogeneity of differentiation, but will vary depending on the cellular composition.[4] Rhabdoid cells, while not present in all AT/RTs, will express vimentin, epithelial membrane antigen, and smooth muscle actin. Immunohistochemistry for the SMARCB1 protein is useful in establishing the diagnosis of AT/RT. A loss of SMARCB1 staining is noted in neoplastic cells, but staining is retained in non-neoplastic cells (e.g., vascular endothelial cells).[5,6,7] AT/RT is a rapidly growing tumor that can have an MIB-1 labeling index of 50% to 100%.[2] Genomics of CNS Atypical Teratoid/Rhabdoid Tumor (AT/RT) SMARCB1gene AT/RT was the first primary pediatric brain tumor for which a candidate tumor suppressor gene, SMARCB1 (also known as INI1 and hSNF5), was identified.[8]SMARCB1 is genomically altered in the majority of rhabdoid tumors, including CNS, renal, and extrarenal rhabdoid malignancies.[8] Additional genomic alterations (mutations and gains/losses) in other genes are very uncommon in patients with SMARCB1-associated AT/RT, and there are no other genes that are recurrently mutated.[9,10,11] SMARCB1 is a component of a switch (SWI) and sucrose non-fermenting (SNF) adenosine triphosphate-dependent chromatin-remodeling complex.[12] Rare familial cases of rhabdoid tumors expressing SMARCB1 and lacking SMARCB1 mutations have also been associated with germline mutations of SMARCA4/BRG1, another member of the SWI/SNF chromatin-remodeling complex.[7,13] The 2016 WHO classification defines AT/RT by the presence of either SMARCB1 or SMARCA4 alterations. Tumors with histological features of AT/RT that lack these genomic alterations are termed CNS embryonal tumor with rhabdoid features.[14] Despite the absence of recurring genomic alterations beyond SMARCB1 (and, more rarely, other SWI/SNF complex members), biologically distinctive subsets of AT/RT have been identified.[15,16] The following three distinctive subsets of AT/RT were identified through the use of DNA methylation arrays for 150 AT/RT tumors and gene expression arrays for 67 AT/RT tumors:[16] - AT/RT TYR: This subset represented approximately one-third of cases and was characterized by elevated expression of melanosomal markers such as TYR (the gene encoding tyrosinase). Cases in this subset were primarily infratentorial, with most presenting in children aged 0 to 1 years and showing chromosome 22q loss.[16] For patients with AT/RT TYR, the mean overall survival (OS) is 37 months (95% confidence interval [CI], 18-56 months).[17] Cribriform neuroepithelial tumor is a brain cancer that also presents in young children and has genomic and epigenomic characteristics that are very similar to AT/RT TYR.[17]
- AT/RT SHH: This subset represented approximately 40% of cases and was characterized by elevated expression of genes in the sonic hedgehog (SHH) pathway (e.g., GLI2 and MYCN). Cases in this subset occurred with similar frequency in the supratentorium and infratentorium. While most presented before age 2 years, approximately one-third of cases presented between ages 2 and 5 years.[16] For patients with AT/RT SHH, the mean OS is 16 months (95% CI, 8-25 months).[17]
- AT/RT MYC: This subset represented approximately one-fourth of cases and was characterized by elevated expression of MYC. AT/RT MYC cases tended to occur in the supratentorial compartment. While most AT/RT MYC cases occurred by age 5 years, AT/RT MYC represented the most common subset diagnosed at age 6 years and older. Focal deletions of SMARCB1 were the most common mechanism of SMARCB1 loss for this subset.[16] For patients with AT/RT MYC, the mean OS is 13 months (95% CI, 5-22 months).[17]
In addition to somatic mutations, germline mutations in SMARCB1 have been reported in a substantial subset of AT/RT patients.[8,18] A study of 65 children with rhabdoid tumors found that 23 (35%) had germline mutations and/or deletions of SMARCB1.[5] Children with germline alterations in SMARCB1 presented at an earlier age than did sporadic cases (median age, approximately 5 months vs. 18 months) and were more likely to present with multiple tumors.[5] One parent was found to be a carrier of the SMARCB1 germline abnormality in 7 of 22 evaluated cases showing germline alterations, with four of the carrier parents being unaffected by SMARCB1-associated cancers.[5] This indicates that AT/RT shows an autosomal dominant inheritance pattern with incomplete penetrance. Gonadal mosaicism has also been observed, as evidenced by families in which multiple siblings are affected by AT/RT and have identical SMARCB1 alterations, but both parents lack a SMARCB1 mutation/deletion.[5,6] Screening children diagnosed with AT/RT for germline SMARCB1 mutations may provide useful information for counseling families on the genetic implications of their child's AT/RT diagnosis.[5] Rhabdoid Tumor Predisposition Syndrome (RTPS) RTPS, related primarily to germline SMARCB1 alterations, has been clearly defined.[8,18] RTPS is highly suggested in patients with synchronous occurrence of renal malignant rhabdoid tumor and AT/RT, bilateral malignant rhabdoid tumors of the kidney, or malignant rhabdoid tumors in two or more siblings. This syndrome is manifested by a marked predisposition to the development of malignant rhabdoid tumors in infancy and early childhood. Up to one-third of AT/RTs are thought to arise in the setting of RTPS, and the majority of these occur within the first year of life. The most common non-CNS malignancy of RTPS is the malignant rhabdoid tumor of the kidney, which is also noted in infancy. For more information about RTPS, refer to the Rhabdoid predisposition syndrome section in the PDQ summary on Wilms Tumor and Other Childhood Kidney Tumors Treatment. References:
-
Rorke LB, Packer RJ, Biegel JA: Central nervous system atypical teratoid/rhabdoid tumors of infancy and childhood: definition of an entity. J Neurosurg 85 (1): 56-65, 1996.
-
Kleihues P, Louis DN, Scheithauer BW, et al.: The WHO classification of tumors of the nervous system. J Neuropathol Exp Neurol 61 (3): 215-25; discussion 226-9, 2002.
-
Kleihues P, Cavenee WK, eds.: Pathology and Genetics of Tumours of the Nervous System. Lyon, France: International Agency for Research on Cancer, 2000.
-
McLendon RE, Adekunle A, Rajaram V, et al.: Embryonal central nervous system neoplasms arising in infants and young children: a pediatric brain tumor consortium study. Arch Pathol Lab Med 135 (8): 984-93, 2011.
-
Eaton KW, Tooke LS, Wainwright LM, et al.: Spectrum of SMARCB1/INI1 mutations in familial and sporadic rhabdoid tumors. Pediatr Blood Cancer 56 (1): 7-15, 2011.
-
Bruggers CS, Bleyl SB, Pysher T, et al.: Clinicopathologic comparison of familial versus sporadic atypical teratoid/rhabdoid tumors (AT/RT) of the central nervous system. Pediatr Blood Cancer 56 (7): 1026-31, 2011.
-
Hasselblatt M, Gesk S, Oyen F, et al.: Nonsense mutation and inactivation of SMARCA4 (BRG1) in an atypical teratoid/rhabdoid tumor showing retained SMARCB1 (INI1) expression. Am J Surg Pathol 35 (6): 933-5, 2011.
-
Biegel JA, Tan L, Zhang F, et al.: Alterations of the hSNF5/INI1 gene in central nervous system atypical teratoid/rhabdoid tumors and renal and extrarenal rhabdoid tumors. Clin Cancer Res 8 (11): 3461-7, 2002.
-
Lee RS, Stewart C, Carter SL, et al.: A remarkably simple genome underlies highly malignant pediatric rhabdoid cancers. J Clin Invest 122 (8): 2983-8, 2012.
-
Kieran MW, Roberts CW, Chi SN, et al.: Absence of oncogenic canonical pathway mutations in aggressive pediatric rhabdoid tumors. Pediatr Blood Cancer 59 (7): 1155-7, 2012.
-
Hasselblatt M, Isken S, Linge A, et al.: High-resolution genomic analysis suggests the absence of recurrent genomic alterations other than SMARCB1 aberrations in atypical teratoid/rhabdoid tumors. Genes Chromosomes Cancer 52 (2): 185-90, 2013.
-
Biegel JA, Kalpana G, Knudsen ES, et al.: The role of INI1 and the SWI/SNF complex in the development of rhabdoid tumors: meeting summary from the workshop on childhood atypical teratoid/rhabdoid tumors. Cancer Res 62 (1): 323-8, 2002.
-
Schneppenheim R, Frühwald MC, Gesk S, et al.: Germline nonsense mutation and somatic inactivation of SMARCA4/BRG1 in a family with rhabdoid tumor predisposition syndrome. Am J Hum Genet 86 (2): 279-84, 2010.
-
Louis DN, Perry A, Reifenberger G, et al.: The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131 (6): 803-20, 2016.
-
Torchia J, Picard D, Lafay-Cousin L, et al.: Molecular subgroups of atypical teratoid rhabdoid tumours in children: an integrated genomic and clinicopathological analysis. Lancet Oncol 16 (5): 569-82, 2015.
-
Johann PD, Erkek S, Zapatka M, et al.: Atypical Teratoid/Rhabdoid Tumors Are Comprised of Three Epigenetic Subgroups with Distinct Enhancer Landscapes. Cancer Cell 29 (3): 379-93, 2016.
-
Johann PD, Hovestadt V, Thomas C, et al.: Cribriform neuroepithelial tumor: Molecular characterization of a SMARCB1-deficient non-rhabdoid tumor with favorable long-term outcome. Brain Pathol : , 2016.
-
Biegel JA, Fogelgren B, Wainwright LM, et al.: Germline INI1 mutation in a patient with a central nervous system atypical teratoid tumor and renal rhabdoid tumor. Genes Chromosomes Cancer 28 (1): 31-7, 2000.
Stage Information for Childhood CNS Atypical Teratoid / Rhabdoid TumorThere is no defined staging system for childhood central nervous system atypical teratoid/rhabdoid tumor. For treatment purposes, patients are classified as having newly diagnosed or recurrent disease with or without neuraxis dissemination. Treatment for Newly Diagnosed Childhood CNS Atypical Teratoid / Rhabdoid TumorThere is no established standard treatment for children with central nervous system (CNS) atypical teratoid/rhabdoid tumor (AT/RT). Given the highly aggressive nature of the tumor, most patients have been treated with intensive multimodal therapy. However, the young age of most patients limits the extent of treatment, particularly radiation therapy. Treatment options for newly diagnosed CNS AT/RT include the following: - Surgery, chemotherapy, and radiation therapy (multimodal therapy).
Surgery, Chemotherapy, and Radiation Therapy (Multimodal Therapy) The extent of the surgical resection may affect survival. Data from the Central Nervous System Atypical Teratoid/Rhabdoid Tumor Registry (AT/RT Registry) suggest that patients who have had a complete resection may have a longer median survival, although complete surgical resection is often difficult given the invasive nature of the tumor.[1] Chemotherapy has been the main adjuvant therapy for very young children with AT/RT. Cooperative group studies that included children younger than 36 months demonstrated poor survival when treated with standard chemotherapeutic regimens alone.[2] The Children's Cancer Group reported a 2-year event-free survival (EFS) of 14% for 28 children younger than 36 months treated with multiagent chemotherapy.[3] Intensive regimens that utilize varying combinations of high-dose chemotherapy,[4][Level of evidence: 3iA]; [5,6][Level of evidence: 3iiiDi] intrathecal chemotherapy, and radiation therapy have led to prolonged survival for some patients. Thirteen patients in the AT/RT Registry were treated with high-dose chemotherapy with hematopoietic stem cell rescue as part of initial therapy.[1] Four of these patients, two of whom also received radiation, were alive without progressive disease 21.5 to 90 months after diagnosis at last report. Of 15 evaluable children, all younger than 32 months at diagnosis, who were on a chemotherapy Head Start III protocol, 2 survived for more than 47 months.[7][Level of evidence: 3iA] Radiation therapy appears to have a positive impact on survival for AT/RT patients. - Of the 42 patients in the AT/RT Registry, 13 patients (31%) received radiation therapy in addition to chemotherapy as part of their primary therapy.[1] The radiation field was to the primary tumor bed in nine children, and the tumor bed and the craniospinal axis in four children. Their median survival was 48 months, while the median survival of all patients on the registry was 16.75 months.
- A retrospective series of 31 patients with AT/RT from St. Jude Children's Research Hospital in which the 2-year EFS for patients older than 3 years was 78%, considerably better than 11% for patients younger than 3 years.[8] All but one of the surviving patients (seven of eight) in the older group received craniospinal irradiation and intensive chemotherapy with hematopoietic stem cell transplant; only 3 of 22 of the younger patients received any form of radiation therapy, two of whom are disease free.
- In a Surveillance, Epidemiology, and End Results registry review, radiation therapy was associated with improved survival in children younger than 3 years.[9]
Evidence (multimodal therapy): - The Third Intergroup Rhabdomyosarcoma Study (IRS-III) utilized radiation therapy, intrathecal methotrexate, cytarabine, hydrocortisone, and systemic multiagent chemotherapy. The results of small retrospective series were encouraging,[10,11] leading to the first prospective study of multimodality treatment in this group of patients.
- Based on the previous pilot series, a prospective multi-institutional trial was conducted for children with newly diagnosed CNS AT/RT. Treatment was divided into five phases: preirradiation, chemoradiation, consolidation, maintenance, and continuation therapy. Intrathecal chemotherapy was administered, alternating intralumbar and intraventricular routes. Radiation therapy was either focal (54 Gy) or craniospinal (36 Gy, plus primary boost), depending on the child's age and extent of disease at diagnosis.[12]
- Results of the prospective study demonstrated a 2-year progression-free survival of 53% ± 13% and an overall survival of 70% ± 10%, with results most favorable in children who were older, had a gross-total resection, and had no metastatic disease at presentation.
- Six of the eight children without progressive disease at the time of the report had received conformal radiation therapy and two children had received craniospinal radiotherapy; seven children had a gross total resection and only one child had metastatic disease (this child had persistent, stable disease 1.5 years from diagnosis).
Because AT/RT is responsive to radiation therapy, this modality is incorporated into many treatment protocols.[13] Prospective cooperative group clinical trials for AT/RT are greatly needed to better understand how age and extent of therapy affect survival. Treatment Options Under Clinical Evaluation Early-phase therapeutic trials may be available for selected patients. These trials may be available via Children's Oncology Group phase I institutions, the Pediatric Brain Tumor Consortium, or other entities. Information about ongoing clinical trials is available from the NCI website. References:
-
Hilden JM, Meerbaum S, Burger P, et al.: Central nervous system atypical teratoid/rhabdoid tumor: results of therapy in children enrolled in a registry. J Clin Oncol 22 (14): 2877-84, 2004.
-
Packer RJ, Biegel JA, Blaney S, et al.: Atypical teratoid/rhabdoid tumor of the central nervous system: report on workshop. J Pediatr Hematol Oncol 24 (5): 337-42, 2002 Jun-Jul.
-
Geyer JR, Sposto R, Jennings M, et al.: Multiagent chemotherapy and deferred radiotherapy in infants with malignant brain tumors: a report from the Children's Cancer Group. J Clin Oncol 23 (30): 7621-31, 2005.
-
Nicolaides T, Tihan T, Horn B, et al.: High-dose chemotherapy and autologous stem cell rescue for atypical teratoid/rhabdoid tumor of the central nervous system. J Neurooncol 98 (1): 117-23, 2010.
-
Gardner SL, Asgharzadeh S, Green A, et al.: Intensive induction chemotherapy followed by high dose chemotherapy with autologous hematopoietic progenitor cell rescue in young children newly diagnosed with central nervous system atypical teratoid rhabdoid tumors. Pediatr Blood Cancer 51 (2): 235-40, 2008.
-
Finkelstein-Shechter T, Gassas A, Mabbott D, et al.: Atypical teratoid or rhabdoid tumors: improved outcome with high-dose chemotherapy. J Pediatr Hematol Oncol 32 (5): e182-6, 2010.
-
Zaky W, Dhall G, Ji L, et al.: Intensive induction chemotherapy followed by myeloablative chemotherapy with autologous hematopoietic progenitor cell rescue for young children newly-diagnosed with central nervous system atypical teratoid/rhabdoid tumors: the Head Start III experience. Pediatr Blood Cancer 61 (1): 95-101, 2014.
-
Tekautz TM, Fuller CE, Blaney S, et al.: Atypical teratoid/rhabdoid tumors (ATRT): improved survival in children 3 years of age and older with radiation therapy and high-dose alkylator-based chemotherapy. J Clin Oncol 23 (7): 1491-9, 2005.
-
Buscariollo DL, Park HS, Roberts KB, et al.: Survival outcomes in atypical teratoid rhabdoid tumor for patients undergoing radiotherapy in a Surveillance, Epidemiology, and End Results analysis. Cancer 118 (17): 4212-9, 2012.
-
Olson TA, Bayar E, Kosnik E, et al.: Successful treatment of disseminated central nervous system malignant rhabdoid tumor. J Pediatr Hematol Oncol 17 (1): 71-5, 1995.
-
Zimmerman MA, Goumnerova LC, Proctor M, et al.: Continuous remission of newly diagnosed and relapsed central nervous system atypical teratoid/rhabdoid tumor. J Neurooncol 72 (1): 77-84, 2005.
-
Chi SN, Zimmerman MA, Yao X, et al.: Intensive multimodality treatment for children with newly diagnosed CNS atypical teratoid rhabdoid tumor. J Clin Oncol 27 (3): 385-9, 2009.
-
De Amorim Bernstein K, Sethi R, Trofimov A, et al.: Early clinical outcomes using proton radiation for children with central nervous system atypical teratoid rhabdoid tumors. Int J Radiat Oncol Biol Phys 86 (1): 114-20, 2013.
Treatment for Recurrent Childhood CNS Atypical Teratoid / Rhabdoid TumorThere is no standard treatment for recurrent childhood central nervous system atypical teratoid/rhabdoid tumor. Treatment Options Under Clinical Evaluation The following is an example of a national and/or institutional clinical trial that is currently being conducted. Other trials may be available via Children's Oncology Group phase I institutions, the Pediatric Brain Tumor Consortium, or other entities. Information about ongoing clinical trials is available from the NCI website. - NCT02601937 (A Phase 1 Study of the EZH2 Inhibitor Tazemetostat in Pediatric Subjects With Relapsed or Refractory INI1-Negative Tumors or Synovial Sarcoma): Patients with INI1-negative tumors are eligible for targeted treatment with an EZH2 inhibitor. This is a phase I, open-label, dose-escalation, and dose-expansion study with a twice-daily oral dose of tazemetostat.
Current Clinical TrialsCheck the list of NCI-supported cancer clinical trials that are now accepting patients with childhood atypical teratoid/rhabdoid tumor. The list of clinical trials can be further narrowed by location, drug, intervention, and other criteria. General information about clinical trials is also available from the NCI website. Changes to This Summary (04 / 17 / 2017)The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above. Tumor Biology of Childhood Central Nervous System (CNS) Atypical Teratoid/Rhabdoid Tumor (AT/RT) Added text to state that the 2016 World Health Organization classification defines AT/RT by the presence of either SMARCB1 or SMARCA4 alterations. Tumors with histological features of AT/RT that lack these genomic alterations are termed CNS embryonal tumor with rhabdoid features (cited Louis et al. as reference 14). Added text to state that for patients with AT/RT TYR, the mean overall survival (OS) is 37 months. Cribriform neuroepithelial tumor is a brain cancer that also presents in young children and has genomic and epigenomic characteristics that are very similar to AT/RT TYR (cited Johann et al. as reference 17). Added text to state that for patients with AT/RT SHH, the mean OS is 16 months. Added text to state that for patients with AT/RT MYC, the mean OS is 13 months. This summary is written and maintained by the PDQ Pediatric Treatment Editorial Board, which is editorially independent of NCI. The summary reflects an independent review of the literature and does not represent a policy statement of NCI or NIH. More information about summary policies and the role of the PDQ Editorial Boards in maintaining the PDQ summaries can be found on the About This PDQ Summary and PDQ® - NCI's Comprehensive Cancer Database pages. About This PDQ SummaryPurpose of This Summary This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about the treatment of childhood central nervous system atypical teratoid and rhabdoid tumor. It is intended as a resource to inform and assist clinicians who care for cancer patients. It does not provide formal guidelines or recommendations for making health care decisions. Reviewers and Updates This summary is reviewed regularly and updated as necessary by the PDQ Pediatric Treatment Editorial Board, which is editorially independent of the National Cancer Institute (NCI). The summary reflects an independent review of the literature and does not represent a policy statement of NCI or the National Institutes of Health (NIH). Board members review recently published articles each month to determine whether an article should: - be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary. The lead reviewers for Childhood Central Nervous System Atypical Teratoid/Rhabdoid Tumor Treatment are: - Kenneth J. Cohen, MD, MBA (Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Hospital)
- Karen J. Marcus, MD (Dana-Farber Cancer Institute/Boston Children's Hospital)
- Roger J. Packer, MD (Children's National Health System)
- Malcolm A. Smith, MD, PhD (National Cancer Institute)
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website's Email Us. Do not contact the individual Board Members with questions or comments about the summaries. Board members will not respond to individual inquiries. Levels of Evidence Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Pediatric Treatment Editorial Board uses a formal evidence ranking system in developing its level-of-evidence designations. Permission to Use This Summary PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as "NCI's PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary]." The preferred citation for this PDQ summary is: PDQ® Pediatric Treatment Editorial Board. PDQ Childhood Central Nervous System Atypical Teratoid/Rhabdoid Tumor Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/brain/hp/child-cns-atrt-treatment-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389426] Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in Visuals Online, a collection of over 2,000 scientific images. Disclaimer Based on the strength of the available evidence, treatment options may be described as either "standard" or "under clinical evaluation." These classifications should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page. Contact Us More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website's Email Us. Last Revised: 2017-04-17 Last modified on: 8 September 2017
|
|